“Ions” By Jkwchui – Own work (CC BY-SA 3. “Periodic Table Of Elements” By Dmarcus100 – Own work (CC BY-SA 4.0) via Commons WikimediaĢ. “What Is Valence or Valency?” ThoughtCo, Mar. In summary, the key difference between valency and charge is that valency indicates the ability of a chemical element to combine with another chemical element, whereas charge indicates the number of electrons a chemical element gains or removes. Valency gives the reactivity of an atom while charge describes how an atom has reacted. In general, atoms that form positive ions (cations) are. So, their ionic charge is usually the same. On the periodic table, elements in the same group have the same number of valence electrons. The charge depends on the atoms electron configuration and the number of valence electrons. The below infographic summarizes the difference between valency and charge. An ionic charge is a positive or negative charge on an atom. Atoms that lose electrons acquire a positive charge as a result because they are left with. Atom may lose valence electrons quite to obtain a lower shell that contains an octet. Moreover, the value for valency has no plus or minus signs, while the charge has plus sign if the ion has formed by removing electrons and has the minus sign if the atom has gained electrons. 4.10: Ions - Losing and Gaining Electrons is shared under a Public Domain license and was authored, remixed, and/or curated by LibreTexts. So, the key difference between valency and charge is that valency indicates the ability of a chemical element to combine with another chemical element, whereas charge indicates the number of electrons either gained or removed by a chemical element. Valency indicates the reactivity of an atom, while charge indicates how an atom has reacted. (b) Covalent radii of the elements are shown to scale. The atomic radius for the halogens increases down the group as n increases. What is the Difference Between Valency and Charge? 1: (a) The radius of an atom is defined as one-half the distance between the nuclei in a molecule consisting of two identical atoms joined by a covalent bond. the integers of which are equal to the positive electrical charges of the atomic nuclei. The elements in a group have very similar chemical properties, which arise from the number of valence electrons presentthat is. But, if the atom removes an electron, then there is one proton in extra thus, the atom gets +1 charge. Periodic table, in chemistry, the organized array of all the chemical elements in order of increasing atomic number. As you go down the periodic table, it becomes easier to remove an electron from an atom (i.e., IE decreases) because the valence electron is farther away from the nucleus. When an atom gains an electron, there aren’t enough protons in the atom to balance this charge thus, the charge of the atom is -1. Here, if an atom gains electrons, then it gets a negative charge since an electron has a negative charge. However, if an atom has an unstable electron configuration, then it tends to form ions by either gaining or removing electrons. 
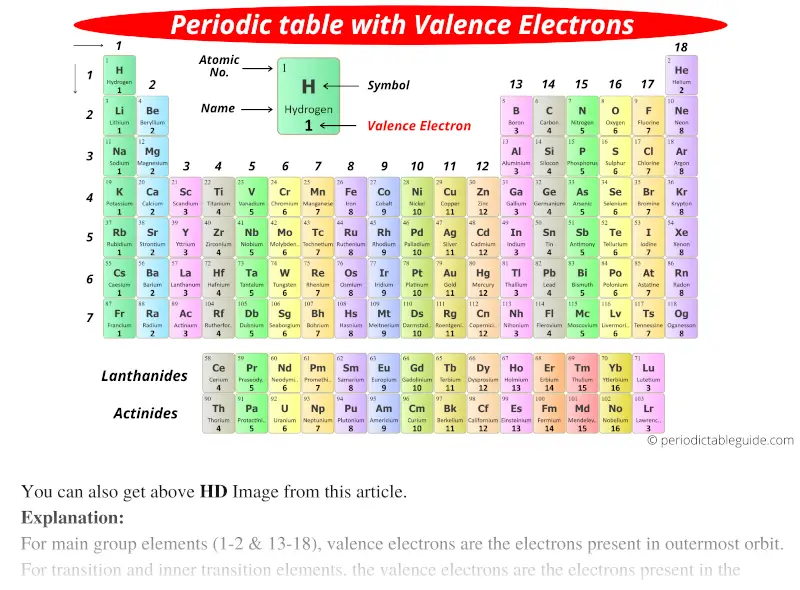
Therefore, they have one electron for the displacement or the combination with a hydrogen atom. For example, group 1 elements in the periodic table have one outermost electron. The number of electrons in the outermost shell determines the valency of the atom as well. The periodic table has arranged the chemical elements according to the number of electrons in the outermost shell of the atom. We can determine the valency by looking at the position of the chemical element in the periodic table. However, it only describes the connectivity of atoms and does not describe the geometry of a compound. It is a measure of the reactivity of a chemical element. Valency is the combining power of an element, especially as measured by the number of hydrogen atoms it can displace or combine with. 
Side by Side Comparison – Valency vs Charge in Tabular Form On the other hand, a charge of an atom is the number of protons minus the number of electrons in an atom. Valency and charge are closely related terms as both these terms describe the reactivity of a chemical element.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
